
For six decades, respiratory syncytial virus (RSV) represented one of the most significant and intractable challenges in pediatric medicine. A primary cause of bronchiolitis and pneumonia in infants, the virus has historically been a seasonal certainty that clinicians could only manage through supportive care rather than prevention. However, the landscape of public health changed fundamentally between 2022 and 2024 with the authorization of long-acting monoclonal antibodies and maternal vaccines within the European Union. This scientific breakthrough has shifted the conversation from a search for medical solutions to a struggle over political will, funding, and structural implementation.
The European Centre for Disease Prevention and Control (ECDC) estimates that approximately 250,000 children under the age of five are hospitalized annually across Europe due to RSV infections. These hospitalizations do more than strain the emotional and financial resources of families; they systematically overwhelm pediatric wards every winter, leading to the deferral of elective surgeries and the exhaustion of healthcare personnel. Despite the availability of new tools, a significant divide has emerged across the continent, separating nations that have successfully integrated these tools into their national health systems from those hindered by political inertia and fragmented delivery mechanisms.
The Evolution of RSV Prevention: A 60-Year Journey
The quest to protect infants from RSV began in the 1960s, but early attempts were met with catastrophic setbacks. A formaldehyd-inactivated vaccine tested in the mid-60s led to enhanced respiratory disease in vaccinated children when they later encountered the wild virus, resulting in severe illness and, in some cases, death. This tragedy stalled RSV vaccine development for decades, as researchers struggled to understand the complex immunology of the virus.

It was not until the discovery of the "pre-fusion" state of the RSV F-protein that a viable path forward emerged. This breakthrough allowed for the development of highly effective preventive measures: nirsevimab, a long-acting monoclonal antibody administered directly to infants, and bivalent maternal vaccines designed to be given during pregnancy to transfer antibodies to the fetus.
By late 2022 and early 2023, the European Medicines Agency (EMA) provided the regulatory green light for these products. Currently, 23 EU/EEA countries have issued recommendations for RSV immunization. However, the transition from recommendation to a fully funded, accessible public health program remains inconsistent across the European landscape.
Real-World Evidence: Successes and Shortfalls
The 2023/24 and 2024/25 RSV seasons provided the first comprehensive "scorecard" for how different national strategies performed under real-world conditions. The data suggests that the choice of preventive modality and the efficiency of the delivery system are the primary determinants of success.
Spain: The Gold Standard of Implementation
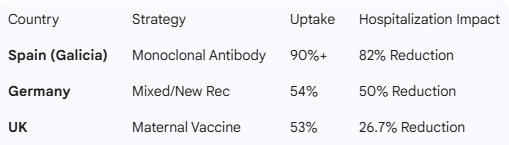
Spain emerged as the European leader in RSV prevention, largely driven by proactive regional health authorities. In the region of Galicia, which was among the first to implement a universal monoclonal antibody program, health officials achieved an uptake rate exceeding 90%. The result was a staggering 82% reduction in infant RSV hospitalizations.

The REACH study, a multi-country analysis presented at the European Society for Paediatric Infectious Diseases (ESPID) in 2025, confirmed these findings on a broader scale. Across Spain, infant RSV hospitalizations dropped by 69% in the 2024/25 season compared to the pre-immunization benchmark of 2022/23. The Spanish model demonstrated that when clinical evidence is paired with high public trust and streamlined delivery—often administering the dose in the birth center—the impact on the healthcare system is immediate and profound.
Italy: Rapid Scale-Up and Regional Coordination
Italy followed a similar trajectory, demonstrating that rapid scale-up is possible even in decentralized health systems. Central recommendations provided the necessary political coverage for regional health authorities to act. In Lombardy, the 2024/25 campaign saw coverage rates rise above 85%, leading to marked reductions in both emergency department visits and inpatient admissions. Italy’s success was attributed to a phased approach, initially focusing on infants born during the height of the RSV season before expanding to a universal cohort.
Germany: Progress Amidst Information Gaps
Germany’s experience under the Standing Committee on Vaccination (STIKO) recommendations showed promising but incomplete results. In the first season of implementation, coverage reached 54% for pre-season infants. While this led to RSV incidence in children under one year dropping by more than half, a significant portion of the population remained unprotected.
A study conducted at the University Hospital of Würzburg found that while only 14% of parents actively declined the intervention, nearly one-third were undecided. This suggests that the primary barrier in Germany is not ideological opposition or "vaccine hesitancy" in the traditional sense, but rather an information gap. Notably, during that season, every severe RSV case requiring pediatric intensive care at the facility occurred in an unimmunized infant, highlighting the high stakes of the communication failure.

The United Kingdom: A Cautionary Tale of Single-Modality Strategy
The United Kingdom opted for a strategy primarily centered on maternal vaccination. While maternal vaccines are a vital tool, the UK’s results lagged behind the monoclonal antibody programs of Spain and Italy. The REACH study measured only a 26.7% reduction in infant RSV hospitalizations in the UK, compared to Spain’s 69%.
Furthermore, the UK program faced significant equity challenges. Maternal vaccine uptake reached 53% overall, but a granular look at the data revealed deep disparities: coverage among Black British-Caribbean women was as low as 28%, compared to 61% among White British women. This highlights the risk of relying on a single delivery pathway that may not be equally accessible to all socioeconomic or ethnic groups.
Structural and Political Barriers to Universal Coverage
The primary obstacles to universal RSV prevention are no longer scientific; they are structural, political, and communicative. One of the most significant hurdles is the framing of RSV within the halls of government. For too long, RSV has been viewed strictly as a pediatric clinical issue. To move the needle on funding, advocates argue that it must be reframed as a population-level public health and workforce issue.
When RSV is framed as a health system capacity problem—one that causes emergency department congestion and forces the cancellation of elective surgeries—it gains the attention of finance and health ministers. Furthermore, the economic impact on the workforce is substantial; when an infant is hospitalized, parents are removed from the workforce, leading to productivity losses that ripple through the economy.
Infrastructural gaps also play a role. In the Netherlands, where home births are common, midwives are the primary points of contact for newborns. However, in many jurisdictions, midwives are not authorized to administer monoclonal antibodies. This creates a "vulnerability window" where an infant may go days or weeks without protection before seeing a general practitioner or visiting a health center.
Terminology has also proven to be a stumbling block. The technical distinction between a "vaccine" (which stimulates the immune system to produce antibodies) and a "monoclonal antibody" (which provides passive immunity directly) is often lost on the public. In a post-COVID-19 environment where vaccine discourse is highly charged, some countries have found greater success by using the simple, umbrella term "RSV prevention."
The Synergistic Threat of Pneumococcal Disease
While RSV is a primary driver of infant hospitalization, it does not act in isolation. Respiratory viruses often pave the way for secondary bacterial infections, most notably pneumococcal disease caused by Streptococcus pneumoniae. Pneumococcal disease remains a leading cause of bacterial pneumonia, meningitis, and sepsis in children globally.
The integration of RSV prevention into broader immunization policies offers a "double dividend." By reducing the incidence of RSV, health systems can also potentially lower the burden of secondary bacterial complications. Strengthening pneumococcal conjugate vaccine (PCV) programs alongside new RSV tools represents a holistic approach to pediatric respiratory health. This synergy is essential for achieving the goals of the Immunization Agenda 2030, which aims to ensure that everyone, everywhere, at every age, benefits from vaccines for good health and well-being.

A Roadmap for 2026/27: Five Strategic Priorities
As Europe prepares for future respiratory seasons, public health experts and former policymakers have identified five essential priorities to ensure that scientific breakthroughs translate into lives saved.
- Mandatory Funding and Universal Access: Every EU/EEA country must establish a funded RSV infant immunization program. Recommendations without financial backing are insufficient. Funding should be integrated into public health budgets or mandatory health insurance schemes to ensure that cost is not a barrier for families.
- Unified and Simplified Messaging: Health authorities should adopt the term "RSV prevention" to reduce confusion. Communication strategies must focus on the immediate benefits: fewer hospital stays, healthier infants, and less strain on the family unit.
- Tailored Delivery Pathways: One size does not fit all. Countries must design delivery systems that reflect their unique healthcare landscapes. For nations with high home-birth rates, this may mean authorizing midwives to provide immunizations. For others, it may involve integrating RSV prevention into routine newborn screenings or the first postnatal check-up.
- Investment in Advocacy Infrastructure: Clinicians and researchers need support to communicate effectively with policymakers. Translating complex clinical data into "politically compelling" arguments—such as hospital beds freed and money saved—is a specialized skill that must be cultivated within the public health community.
- Transparent and Rapid Data Sharing: The success of the Spanish model was bolstered by the rapid publication of results in international, English-language journals. Every country implementing these programs has a responsibility to contribute to the global evidence base, allowing others to learn from their successes and failures in real-time.
Conclusion: The Urgency of Political Courage
The scientific community has fulfilled its mandate by delivering safe and effective tools to combat a virus that has plagued infants for generations. The burden of responsibility has now shifted to the political arena. Every winter that passes without comprehensive RSV and pneumococcal prevention policies is a season characterized by preventable suffering, avoidable hospitalizations, and unnecessary economic strain.
The evidence from Spain, Italy, and Germany proves that the tools work and the public is willing to accept them when provided with clear information and easy access. As Europe moves toward the 2026/27 season, the challenge is to bridge the gap between scientific possibility and political reality. We have waited sixty years for these medical advancements; to let them sit on the shelf due to administrative or political hesitation would be a failure of public health leadership. The time for "breathing new life" into immunization policies is now.


